Ophthalmology
Browse to: Geographic Atrophy | Thyroid Eye Disease
Geographic Atrophy
Geographic Atrophy, the advanced form of dry age-related macular degeneration (Dry AMD), affects approximately 2 million people in the United States and European Union. Illustrating the profound societal impact of this disease, approximately 20 percent of cases of legal blindness in North America are caused by Geographic Atrophy.
Patients with Geographic Atrophy suffer from chronic deterioration of cells in the retina, an inner layer of the eye critical for sight, resulting in progressive and irreversible vision loss. Geographic Atrophy can cause blind spots that impair patients’ ability to recognize faces, read, and drive, severely limiting their independence and quality of life.
Existing treatments require monthly or bimonthly physician-administered intravitreal injections that can be burdensome for patients.
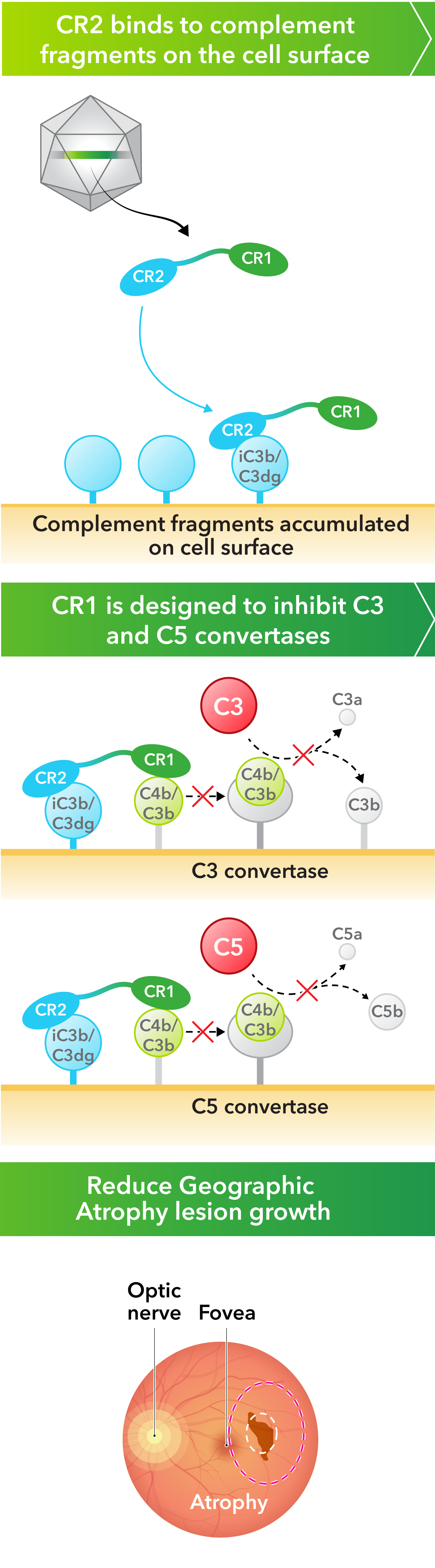
Kriya is developing KRIYA-825, a potential one-time gene therapy for Geographic Atrophy that expresses a fusion protein designed to inhibit the activity of complement C3 and C5, with the objective of slowing Geographic Atrophy lesion growth and vision loss. KRIYA-825 is designed to be administered through a one-time in-office suprachoroidal injection.
Kriya has designed KRIYA‑825 for Geographic Atrophy with the following potential goals in mind:
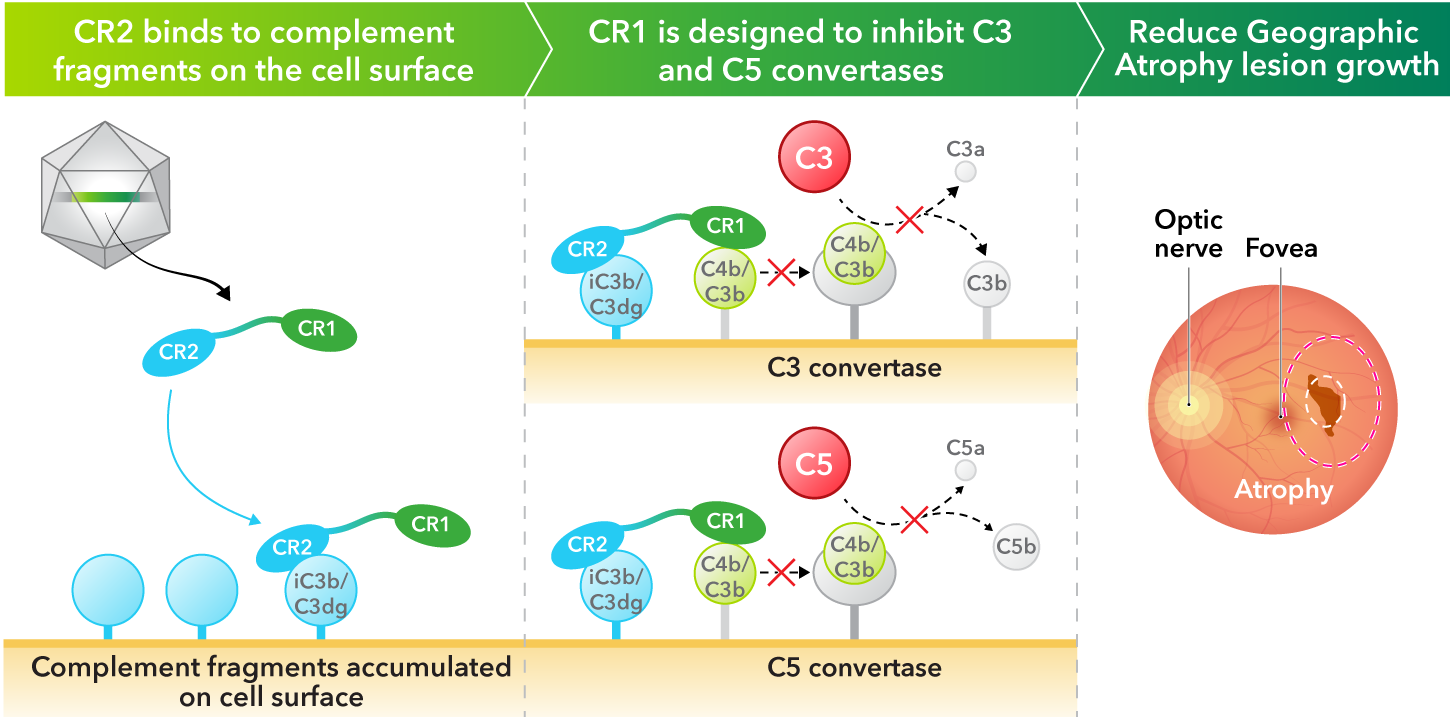
- Robust complement inhibition. A novel complement receptor 2-complement receptor 1 (CR2-CR1) fusion protein—where the CR1 domain is designed to block the activity of both complement C3 and C5, while the CR2 domain is designed to bind to the surfaces of cells where complement fragments deposit and cause damage
- Multi-year durability. AAV mediated continuous expression of CR2-CR1 fusion protein following a one-time injection to eliminate the need for frequent intravitreal injections as required by currently available therapies for Geographic Atrophy
- Targeted delivery. One-time suprachoroidal injection to achieve transduction of, and delivery of therapeutic protein to, retinal cells while minimizing inflammation and overall patient burden.

Approach to Treating Geographic Atrophy with Gene Therapy
Kriya’s AAV gene therapy encodes a fusion protein that combines critical domains of complement receptor 2 (CR2) and complement receptor 1 (CR1). CR1 is designed to inhibit the C3 and C5 pathways in the complement cascade, while CR2 prolongs and enhances the inhibitory effect of CR1 by binding the protein to complement fragments deposited on the cell surface, where they accumulate and cause damage. Taken together, the AAV-expressed CR2‑CR1 fusion protein may provide multiyear therapeutic benefit following a one-time injection.
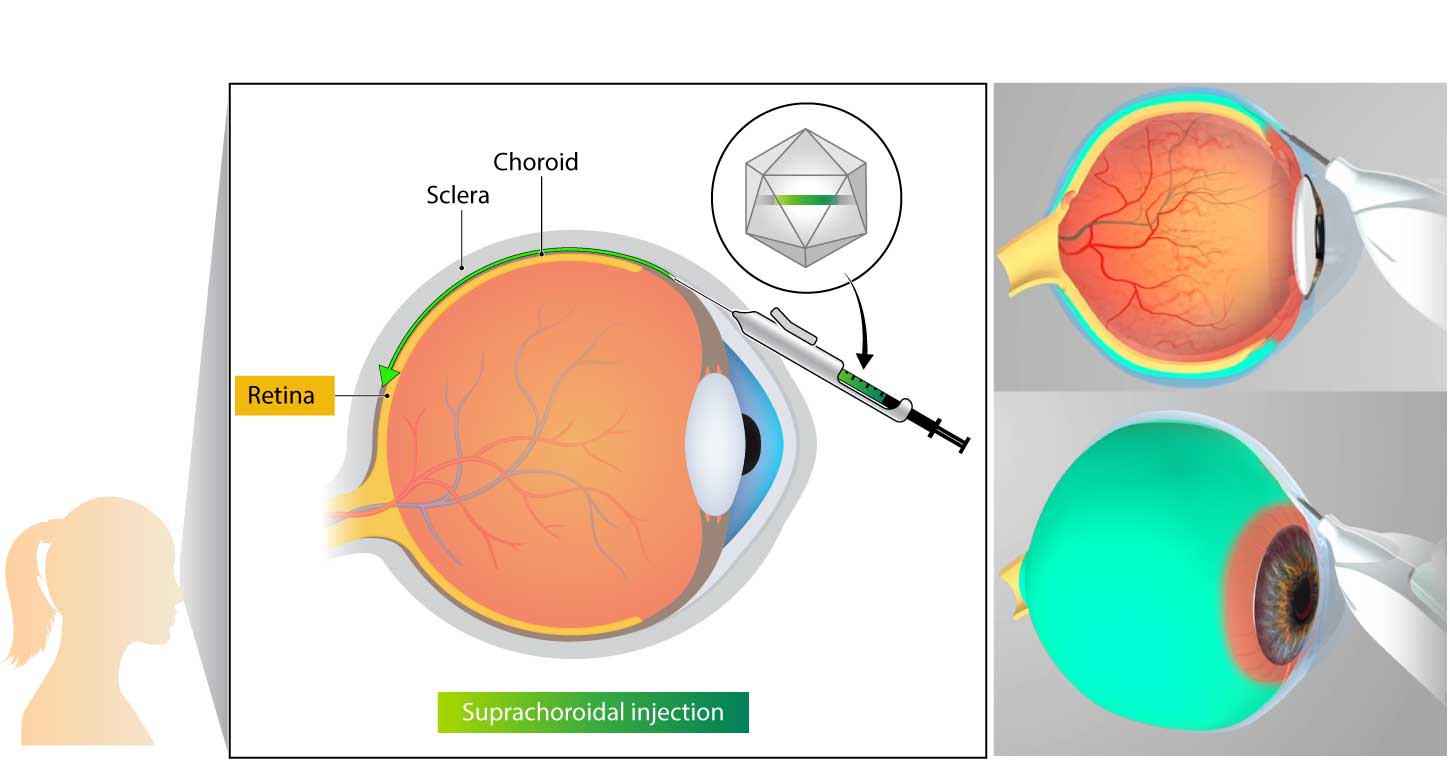
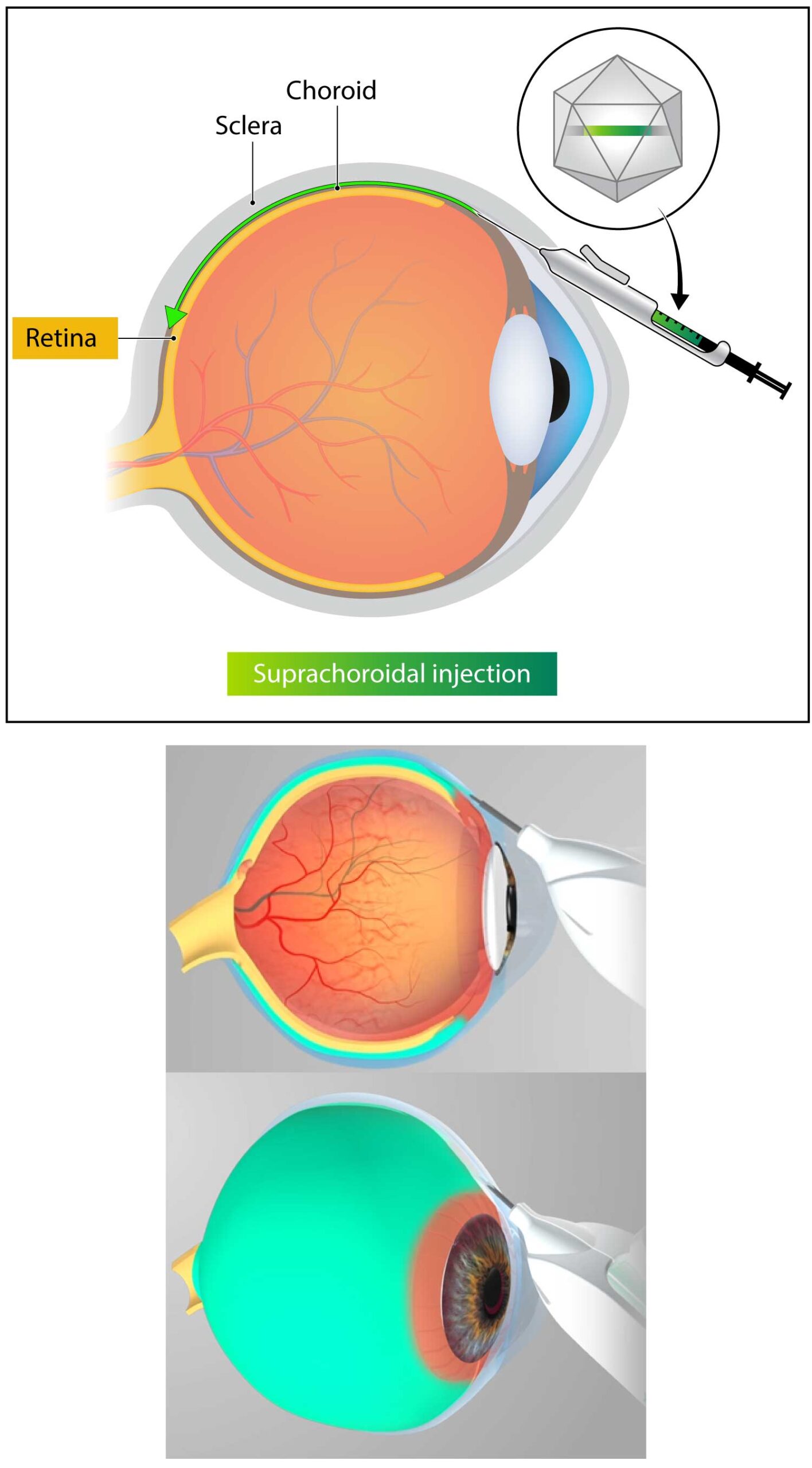
Kriya uses an investigational suprachoroidal delivery device that enables non-surgical targeted delivery of its gene therapy to the retina and choroid via the suprachoroidal space. The delivery device utilizes a geometrically-optimized, non-sharp tissue separator to open a path into the suprachoroidal space, enabling a tangential injection that leads to rapid, broad distribution of the drug into the retina.
Thyroid Eye Disease (TED)
TED is an autoimmune disease that causes inflammation and enlargement of the muscle and fat tissue behind the eye. This results in proptosis (bulging of the eyes) and diplopia (double vision) which can significantly impact quality of life of patients with TED. The only FDA approved therapy for the treatment of TED is an intravenously administered monoclonal antibody that works by blocking Insulin-Like Growth Factor 1 Receptor (IGF1R). There is a need for safe and durable treatments that can address the approximately one million patients in the United States and the European Union who have TED.
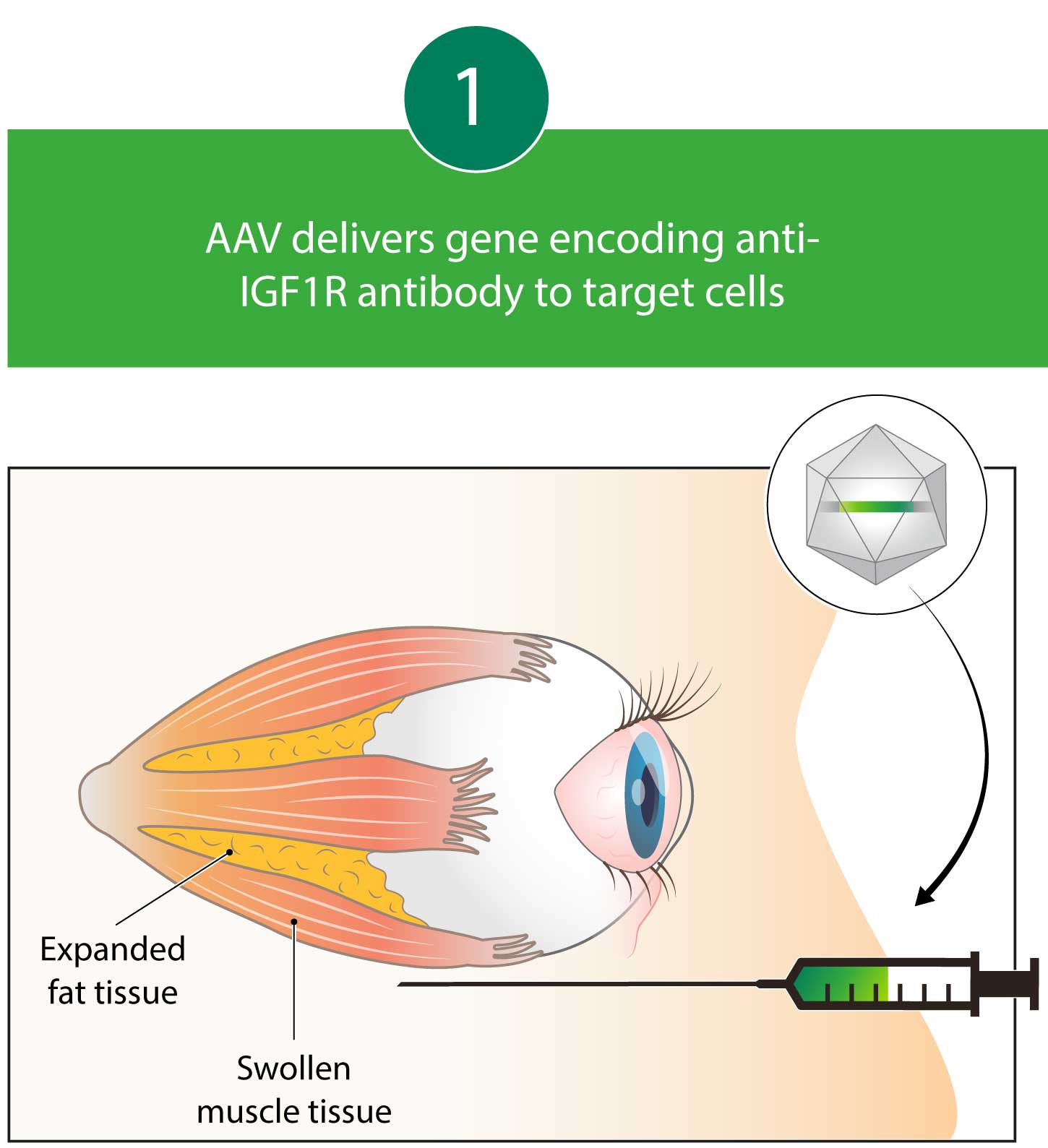
Kriya is developing KRIYA-586, a potential one-time gene therapy for TED engineered to express an antibody that inhibits IGF1R, a key target implicated in the pathogenesis of TED. Currently, the only medicine approved in the U.S. for the treatment of TED is an intravenously infused human monoclonal antibody that blocks IGF1R. KRIYA-586 is designed for one-time delivery through a focal peribulbar injection performed in the office—with the goal of providing local and durable expression of anti-IGF1R antibody from transduced periorbital fat and muscle cells to help address the ocular manifestations of TED while minimizing systemic toxicities.
Kriya has designed KRIYA-586 for TED with the following potential goals in mind:
- One-time administration: One-time, in-office peribulbar injection to eliminate the burdensome requirement for multiple intravenous infusions.
- Focal delivery: Localized antibody expression in extraocular muscles and fat tissue, with low systemic exposure to limit the potential for side effects.
- Long-term durability: AAV-mediated anti-IGF1R expression to deliver sustained improvements in proptosis and diplopia as well as other key manifestations of TED.

Approach to Treating Thyroid Eye Disease with Gene Therapy
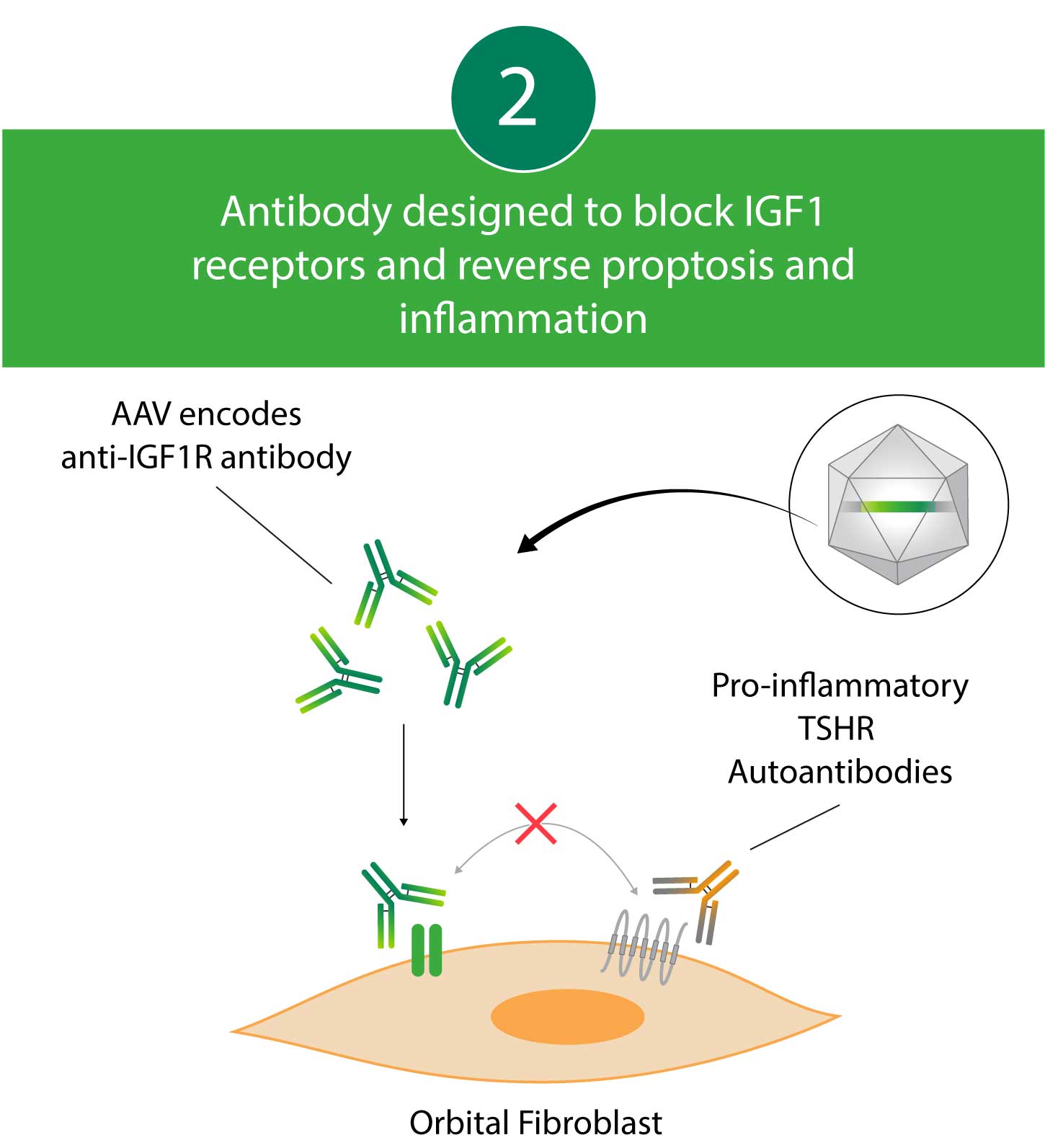
Kriya’s AAV gene therapy encodes an antibody that blocks Insulin-like Growth Factor 1 (IGF1) receptors. By blocking IGF1 receptors, this antibody decreases pro-inflammatory Thyrotropin Receptor (TSHR) autoantibody signaling, which may lead to reductions in proptosis and diplopia. Kriya’s gene therapy is designed for administration as a one-time, focal, peribulbar injection into fatty tissue around the eye, with a goal of achieving local antibody expression and minimizing systemic exposure.